Environmental Implications of Nanotechnology
Funding: National Science Foundation, Virginia Tech Institute of Critical Technology and Applied Science, US Environmental Protection Agency, NSF-EPA Center for the Environmental Implications of Nanotechnology (CEINT)
Our work in this area examine the effects of solution chemistry on the aggregation and dissolution of environmentally relevant nanomaterials. Using four model particle types (gold, silver, C60, and magnetite [Fe3O4]) we have developed a fundamental understanding of the complexities of these systems.
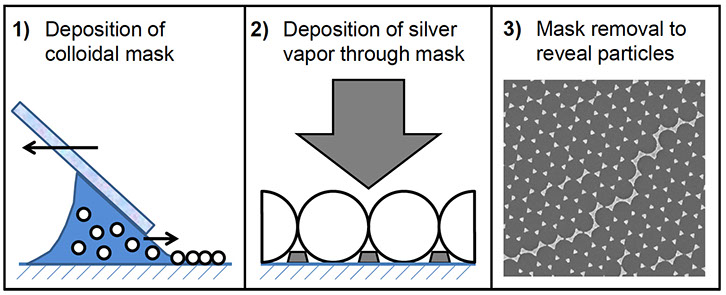
Nanoparticle Transformations
A key focus of the Vikesland research group has been the characterization of nanoparticle transformation processes. One way that we have achieved this goal has been through the development of a highly novel approach that relies upon the use of an atomic force microscope to characterize nanoscale transformations of silver and other labile nanomaterials. Via this approach we are able to quantify transformation kinetics in the absence of artifacts that might arise due to nanoparticle aggregation or the use of non-environmentally relevant nanoparticle concentrations.
Reference publication: Kent, R.D. and Vikesland, P.J. (2012). “Controlled Evaluation of Silver Nanoparticle Dissolution Using Atomic Force Microscopy” Environmental Science and Technology, Vol. 46, pp. 6977-6984.
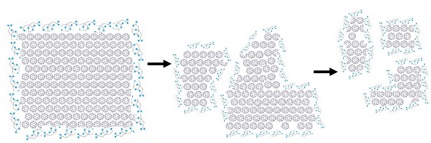
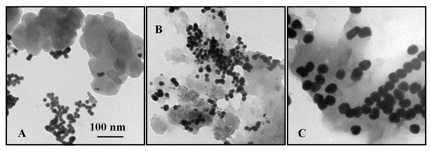
Past studies conducted by our group and others have shown that the association of natural organic matter (NOM) with a nanomaterial surface can either colloidally stabilize the nanomaterial or enhance its dissolution. To probe these effects, we have taken a reductionist approach to evaluate how carboxyl groups affect both the collodial and the chemical stability of nanomaterials. For example in the presence of citrate large aggregates of C60 dissolve to produce small, highly regular cit-nC60 nanoparticles.
Reference publication: Chang, X.; Duncan, L.K.; Jinschek, J.; and Vikesland, P.J. (2012). “Alteration of nC60 in the Presence of Environmentally Relevant Carboxylates” Langmuir, Vol. 28, pp. 7622-7630.
Dr. Peter Vikesland
1145 Perry Street (0246)
415 Durham Hall
Virginia Tech
Blacksburg, VA 24061
Vikesland Research Group
E-mail: pvikes 'at' vt edu
(540) 231-3568